Delving into the history of the atom is like embarking on a fascinating journey through scientific discovery. From ancient philosophical musings to modern quantum mechanics, the understanding of the atom has evolved dramatically. A “History of the Atom” worksheet is a fantastic tool for students to grasp this complex timeline. It typically covers key milestones and the scientists who contributed to our current atomic model. These worksheets often include questions about the contributions of figures like Democritus, Dalton, Thomson, Rutherford, Bohr, and Schrödinger. They may also address key experiments like the cathode ray tube experiment and the gold foil experiment, which provided crucial evidence for the existence of subatomic particles and the structure of the atom. Working through these worksheets encourages critical thinking, research skills, and a deeper appreciation for the scientific method.
Key Figures in Atomic History
Understanding the evolution of the atomic model requires recognizing the contributions of several pivotal scientists. Each scientist built upon the work of their predecessors, refining our understanding of the atom’s composition and behavior. The “History of the Atom” worksheet often focuses on the progression of these ideas and the evidence that supported each new model.
From Philosophy to Science: Democritus and Dalton
The story begins with Democritus, an ancient Greek philosopher who proposed the concept of “atomos,” meaning indivisible. While his idea was purely philosophical and lacked experimental evidence, it laid the groundwork for future atomic theories. Centuries later, John Dalton, an English chemist, revived the atomic theory based on experimental observations. Dalton’s atomic theory, presented in the early 19th century, stated that elements are composed of indivisible atoms, atoms of a given element are identical, and chemical reactions involve the rearrangement of atoms. His work marked a significant shift from philosophical speculation to scientific inquiry.
Discovering Subatomic Particles: Thomson and Rutherford
The notion of the atom as an indivisible entity was shattered with J.J. Thomson’s discovery of the electron in 1897. Using the cathode ray tube experiment, Thomson demonstrated that atoms contained negatively charged particles, later named electrons. He proposed the “plum pudding model,” where electrons were embedded in a positively charged sphere. Ernest Rutherford, a student of Thomson, further revolutionized atomic theory with his gold foil experiment. By bombarding a thin gold foil with alpha particles, Rutherford discovered that most of the alpha particles passed through undeflected, while a small fraction was deflected at large angles. This led him to conclude that the atom consists of a small, dense, positively charged nucleus surrounded by mostly empty space where electrons reside. This model replaced the “plum pudding model” and laid the foundation for the nuclear model of the atom.
Quantization and Beyond: Bohr and Schrödinger
Rutherford’s model, however, had its limitations. It couldn’t explain the stability of the atom or the discrete emission spectra of elements. Niels Bohr addressed these issues by proposing that electrons orbit the nucleus in specific energy levels or shells. Electrons could only occupy these allowed orbits, and transitions between energy levels resulted in the emission or absorption of specific wavelengths of light. This model successfully explained the hydrogen atom’s spectrum but struggled to explain more complex atoms. The development of quantum mechanics in the 20th century brought about a more sophisticated understanding of the atom. Erwin Schrödinger formulated the Schrödinger equation, which describes the wave-like behavior of electrons and provides a probabilistic description of electron location within the atom. This led to the development of the modern quantum mechanical model of the atom, where electrons are described by atomic orbitals, regions of space where there is a high probability of finding an electron.
By completing a “History of the Atom” worksheet, students gain a comprehensive understanding of the key scientific advancements and the scientists who shaped our understanding of the atom. It allows them to appreciate the iterative nature of scientific progress and the importance of experimental evidence in driving scientific theories.
Answers to Common History of the Atom Worksheet Questions
Here are some potential answers to questions that might appear in a typical “History of the Atom” worksheet. Note that the specifics may vary depending on the curriculum and the focus of the worksheet.
- Who first proposed the idea of atoms?
- Democritus (and Leucippus)
- What was Dalton’s atomic theory?
- All matter is composed of atoms.
- Atoms of a given element are identical in size, mass, and other properties.
- Atoms cannot be subdivided, created, or destroyed.
- Atoms of different elements can combine in simple whole number ratios to form chemical compounds.
- In chemical reactions, atoms are combined, separated, or rearranged.
- Describe Thomson’s experiment and his conclusions.
- Experiment: Cathode Ray Tube experiment.
- Conclusions: Atoms contain negatively charged particles (electrons), which are much smaller than the atom itself. He proposed the “plum pudding” model.
- Describe Rutherford’s experiment and his conclusions.
- Experiment: Gold Foil experiment.
- Conclusions: Atoms have a small, dense, positively charged nucleus surrounded by mostly empty space.
- What was Bohr’s model of the atom?
- Electrons orbit the nucleus in specific energy levels or shells.
- Electrons can only occupy these allowed orbits.
- Transitions between energy levels result in the emission or absorption of specific wavelengths of light.
- What is the modern quantum mechanical model of the atom?
- Electrons are described by atomic orbitals, regions of space where there is a high probability of finding an electron.
- Electron behavior is described by the Schrödinger equation, which treats electrons as waves.
- How did Rutherford’s model differ from Thomson’s model?
- Thomson’s model (“plum pudding”) had electrons embedded in a positively charged sphere.
- Rutherford’s model had a small, dense, positively charged nucleus surrounded by mostly empty space with electrons orbiting around it.
- What evidence supported Bohr’s model?
- The discrete emission spectra of elements, particularly the hydrogen atom.
- In the modern atomic model, what determines the properties of an atom?
- The number of protons (atomic number) in the nucleus, as well as the arrangement and number of electrons.
If you are looking for Before we destroy our democracy, we better study history, By 'Tope Fasua you’ve visit to the right page. We have 20 Pics about Before we destroy our democracy, we better study history, By 'Tope Fasua like History, US history timeline and also The History of Data: From Ancient Times to Modern Day | 365 Data Science. Here you go:
Before We Destroy Our Democracy, We Better Study History, By 'Tope Fasua

www.premiumtimesng.com
Understanding History A Primer Of Historical Method Pdf – Bytelinda

bytelinda.weebly.com
Historical Black People – 13 Most Famous – Have Fun With History

www.havefunwithhistory.com
What Is History?

alphahistory.com
Facts & Events That Happened Today In History – The Fact Site

www.thefactsite.com
History

snp13.weebly.com
History Vector

ar.inspiredpencil.com
Customize 21+ History Infographics Templates Online – Canva

www.canva.com
US History Timeline

view.genial.ly
History Of Aravidu Dynasty | Mintage World

www.mintageworld.com
Premium Vector | History Doodle Line Set School University Outline

in.pinterest.com
History | Civil Services Preparation Online | UPSC And IAS Study Material

iasmania.com
Connecting The Past With The Present: The Incredible Power Of

www.historythroughfiction.com
The History Of Data: From Ancient Times To Modern Day | 365 Data Science
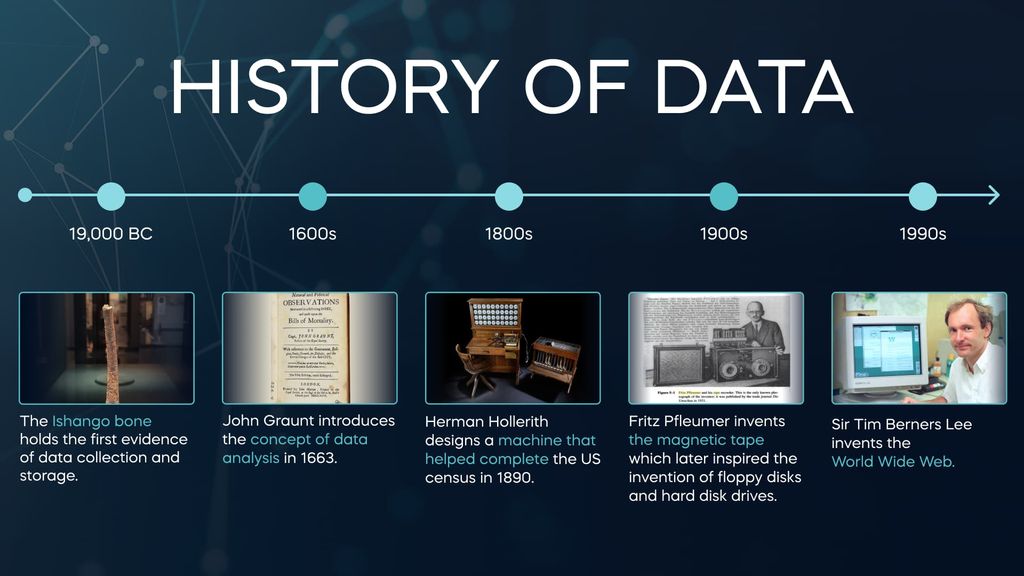
365datascience.com
Esse Texto E Essa Imagem Retratam Um Contexto Marcado

robot.ekstrabladet.dk
History | Poole Grammar School

www.poolegrammar.com
History & Geography To Replace Social Studies As Compulsory Subjects In

way2barak.com
All Humans Have Is The Brief History Of Time, And That Has Been Changed
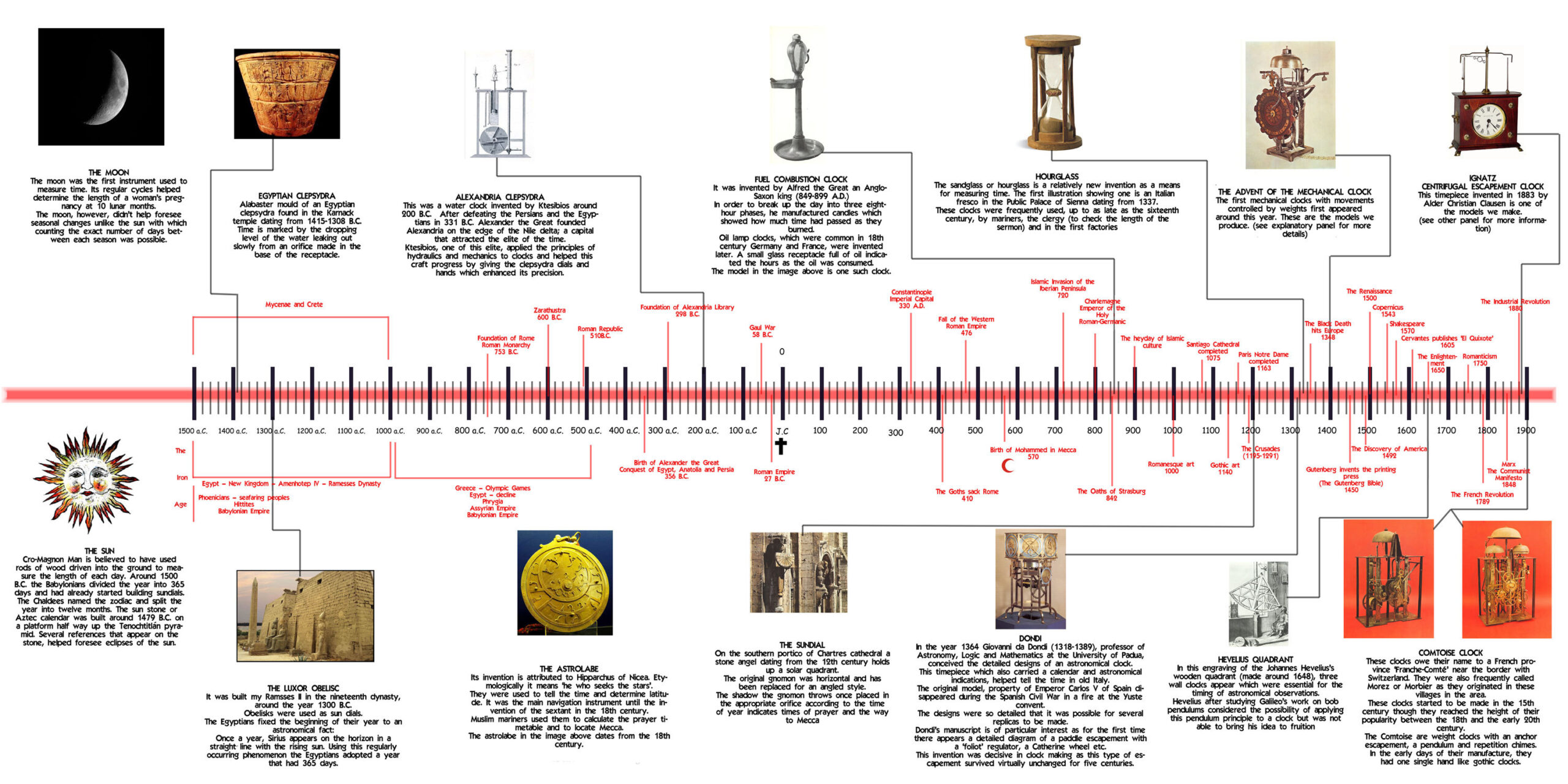
www.theindianwire.com
History | Deparment Of Library & Information Services

uonlibrary.uonbi.ac.ke
What Is History? | 8 Humanities

share.nanjing-school.com
Historical black people. Historical black people. Premium vector