Are you diving into the fascinating world of mixtures? Understanding the differences between solutions, colloids, and suspensions is a cornerstone of chemistry and crucial for grasping everyday phenomena, from the milk you drink to the air you breathe! To solidify your understanding, tackling a “Solutions, Colloids, and Suspensions Worksheet” is a fantastic approach. These worksheets typically involve classifying mixtures based on their properties, identifying examples, and explaining the key distinctions that set them apart. Let’s explore what makes each type of mixture unique and how to effectively tackle these worksheets.
Before we jump into a sample answer key, let’s briefly recap the defining characteristics of each type of mixture:
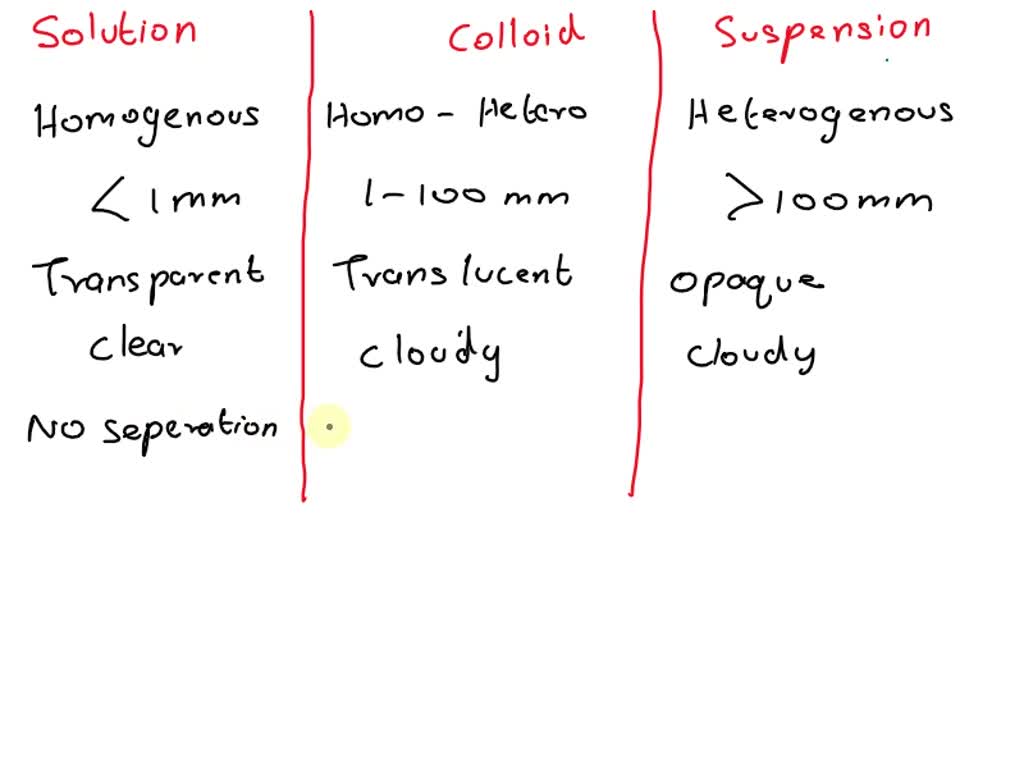
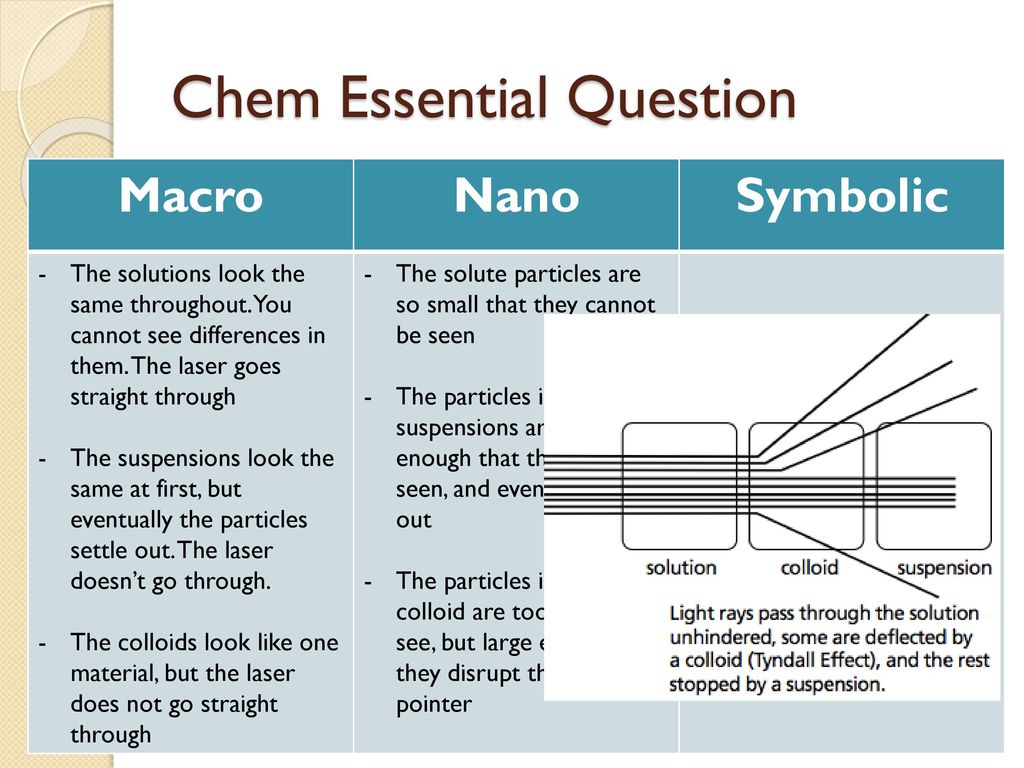
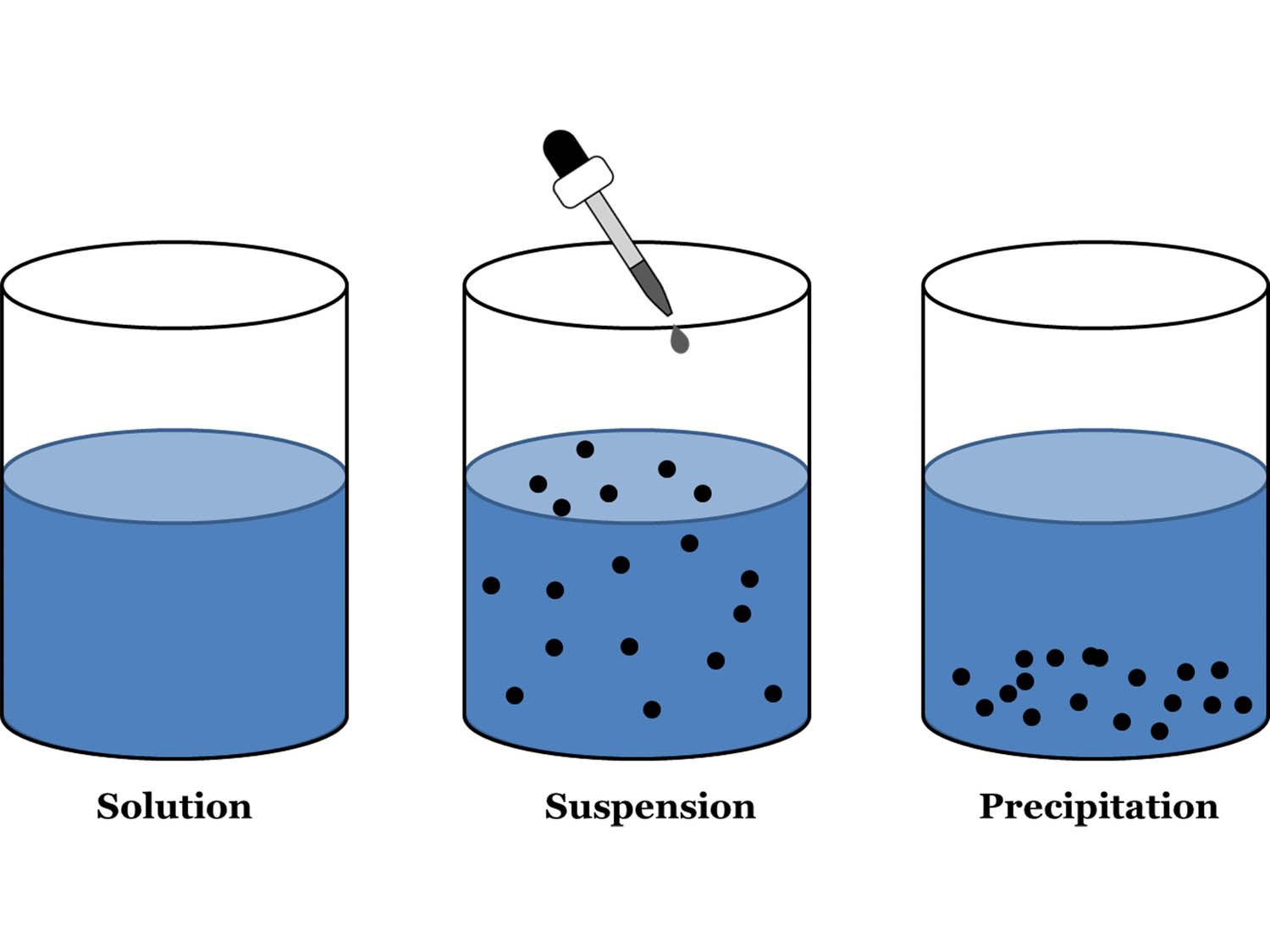
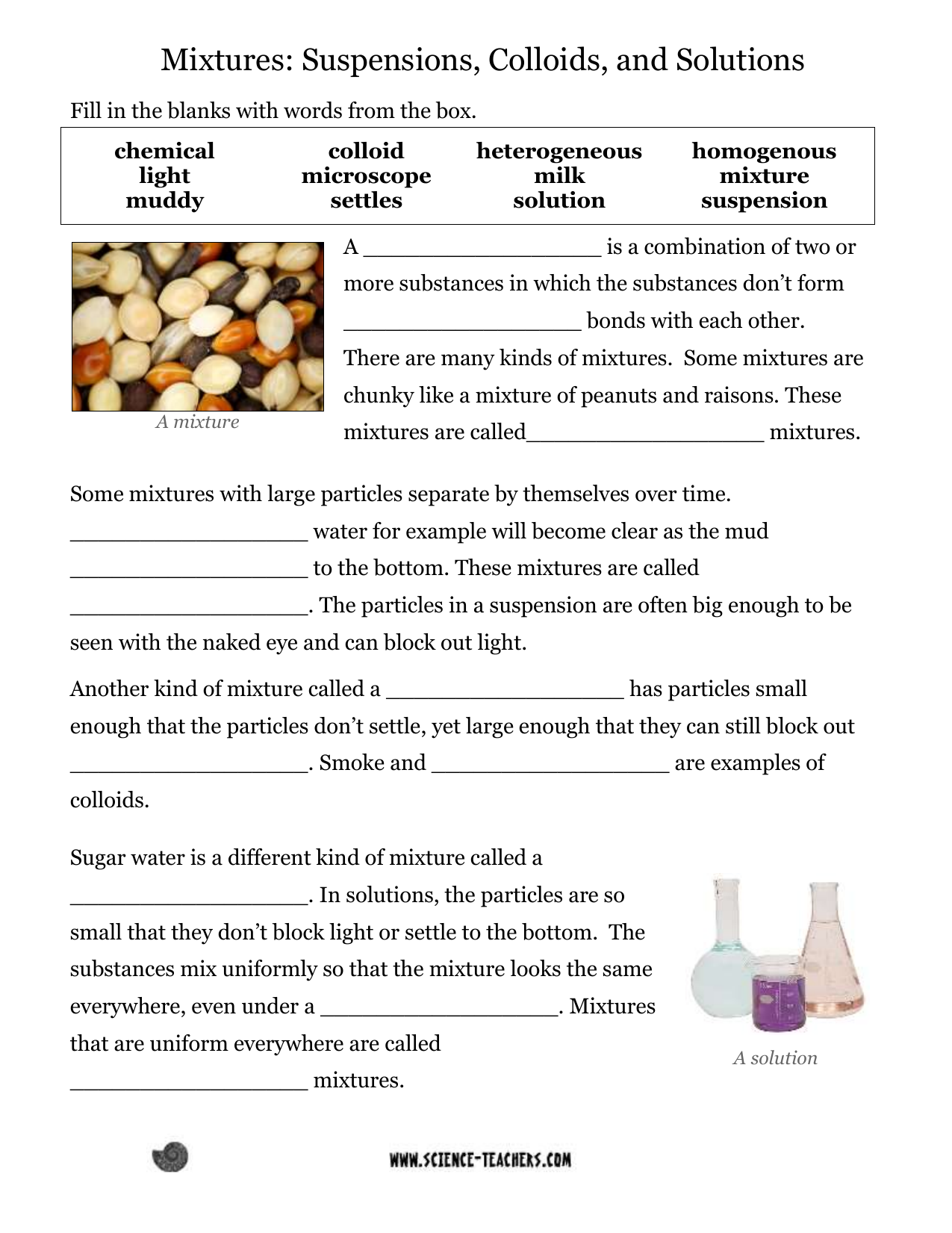
* **Solutions:** These are homogeneous mixtures where one substance (the solute) is uniformly dissolved in another (the solvent). Solutions are transparent, and the solute particles are so small that they cannot be seen even with a microscope. They don’t scatter light (no Tyndall effect), and the solute doesn’t settle out. Examples include saltwater, sugar dissolved in water, and air.
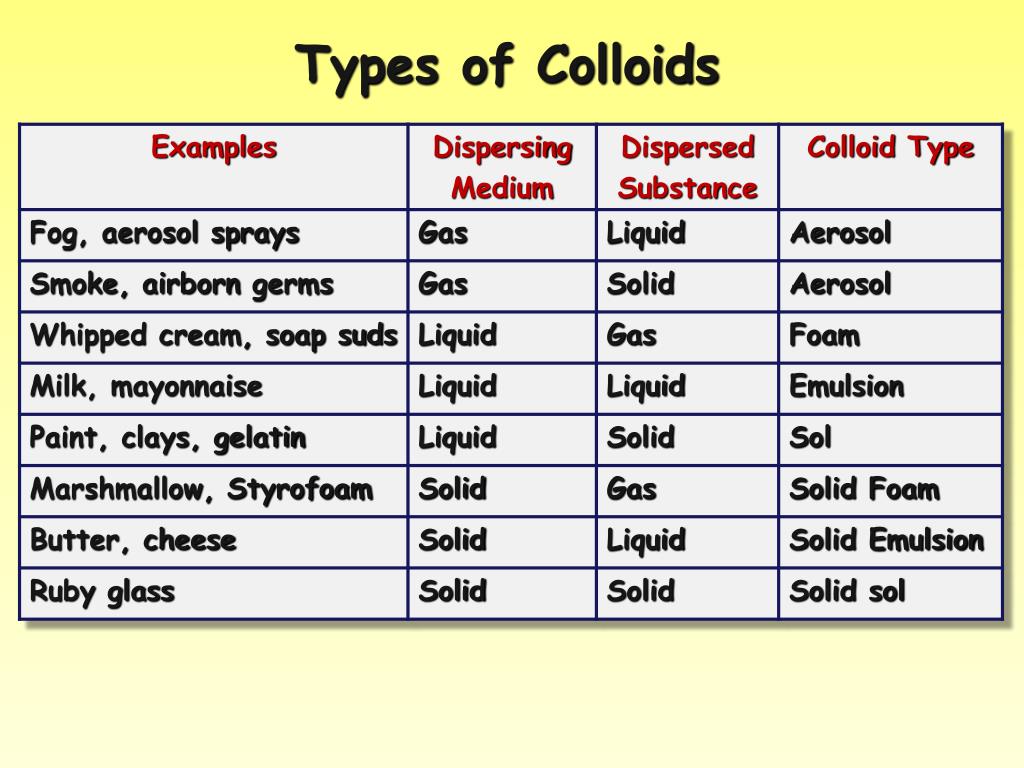
* **Colloids:** Colloids are heterogeneous mixtures where particles are larger than those in solutions but smaller than those in suspensions. They appear cloudy or opaque. A key characteristic of colloids is the Tyndall effect, where light is scattered by the particles. The particles don’t settle out. Examples include milk, fog, and gelatin.
* **Suspensions:** Suspensions are heterogeneous mixtures where particles are large enough to be visible. They are often cloudy or opaque, and the particles will eventually settle out if left undisturbed. They also exhibit the Tyndall effect. Examples include muddy water, sand in water, and paint.
Now, let’s imagine a typical “Solutions, Colloids, and Suspensions Worksheet” and provide potential answers in a helpful format. Keep in mind that the specific questions will vary depending on the worksheet you’re using.
Sample Solutions, Colloids, and Suspensions Worksheet Answer Key
Below are sample answers to common types of questions found on worksheets covering solutions, colloids, and suspensions. Remember to always refer to your specific worksheet instructions and the information provided in your textbook or class notes.
Part 1: Classification
Classify each of the following as a solution, colloid, or suspension:
- Muddy Water
- Saltwater
- Milk
- Fog
- Blood
- Sand in Water
- Vinegar
- Smoke
Here are the corresponding classifications in HTML list format:
- Muddy Water: Suspension
- Saltwater: Solution
- Milk: Colloid
- Fog: Colloid
- Blood: Colloid (specifically, a biological colloid)
- Sand in Water: Suspension
- Vinegar: Solution
- Smoke: Colloid
Part 2: Properties
Fill in the table below to compare the properties of solutions, colloids, and suspensions:
| Property | Solution | Colloid | Suspension |
|---|---|---|---|
| Particle Size | Small (less than 1 nm) | Intermediate (1 nm – 1000 nm) | Large (greater than 1000 nm) |
| Homogeneous/Heterogeneous | Homogeneous | Heterogeneous | Heterogeneous |
| Settles Upon Standing | No | No | Yes |
| Tyndall Effect | No | Yes | Yes |
| Appearance | Transparent | Cloudy/Opaque | Cloudy/Opaque |
Part 3: Explanations
Answer the following questions in complete sentences:
- Explain why saltwater is considered a solution.
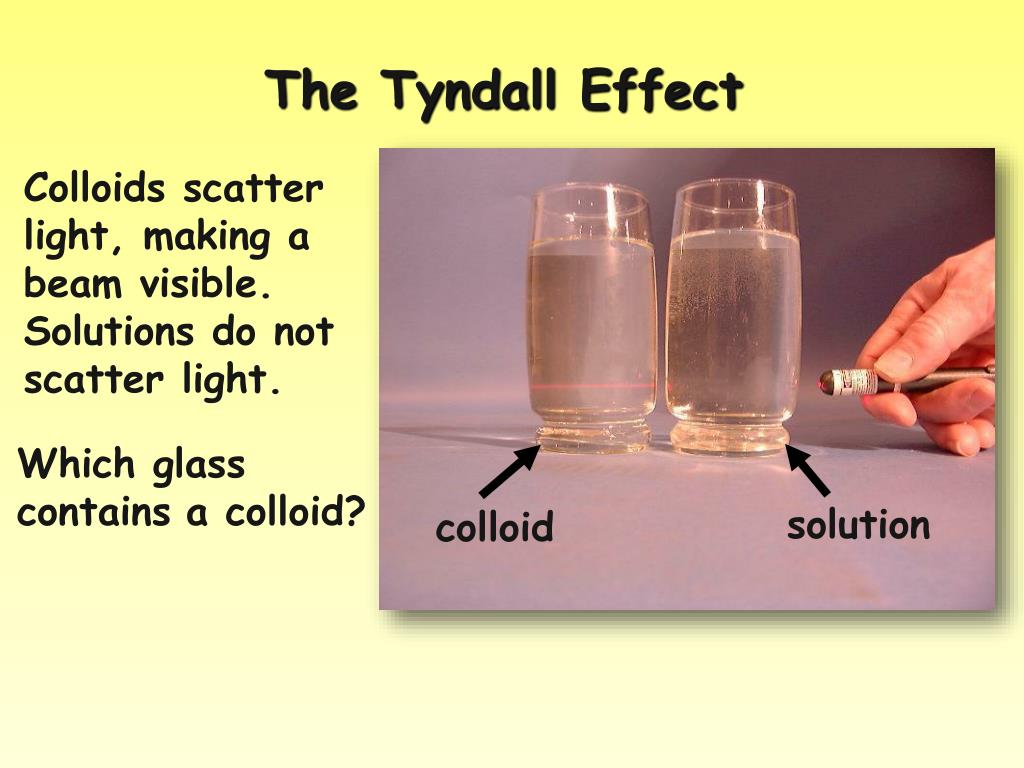
- What is the Tyndall effect, and why does it occur in colloids and suspensions but not in solutions?
- Describe a method you could use to separate sand from a mixture of sand and water. What type of mixture is this?
Here are possible answers to the questions:
- Saltwater is considered a solution because the salt (solute) dissolves completely and uniformly in the water (solvent). The mixture appears transparent, and the salt particles are so small that they are not visible and do not settle out.
- The Tyndall effect is the scattering of light by particles in a mixture. It occurs in colloids and suspensions because the particles are large enough to scatter light beams, making the beam visible. Solutions do not exhibit the Tyndall effect because the solute particles are too small to scatter light effectively.
- To separate sand from a mixture of sand and water, you could use filtration. Pour the mixture through a filter paper placed in a funnel. The water will pass through the filter paper, while the sand will be trapped on the filter paper. This mixture is a suspension because the sand particles are large enough to be visible and will settle out over time.
By carefully considering the properties of each type of mixture – particle size, homogeneity, settling behavior, and the Tyndall effect – you’ll be well-equipped to tackle any “Solutions, Colloids, and Suspensions Worksheet” that comes your way! Good luck, and happy learning!
If you are looking for SOLVED: What are the differences between solutions, colloids, and you’ve came to the right place. We have 20 Pics about SOLVED: What are the differences between solutions, colloids, and like Solutions Colloids And Suspensions Worksheet – Printable PDF Template, Solutions Colloids And Suspensions Worksheet – Ame.my.id and also GRADE 6 CONCEPTS – (NSC) by Janice Bowes-Wellington: MIXTURES. Here you go:
SOLVED: What Are The Differences Between Solutions, Colloids, And
www.numerade.com
Solutions Colloids And Suspensions Worksheet – Owhentheyanks.com

www.owhentheyanks.com
Solutions Colloids And Suspensions Worksheet – Owhentheyanks.com

www.owhentheyanks.com
Solutions Suspensions And Colloids Worksheet – Printable Calendars AT A

ataglance.randstad.com
GRADE 6 CONCEPTS – (NSC) By Janice Bowes-Wellington: MIXTURES

janicebowes70.blogspot.com
GRADE 6 CONCEPTS – (NSC) By Janice Bowes-Wellington: MIXTURES

janicebowes70.blogspot.com
SOLUTION: Worksheet Balancing Equations – Studypool – Worksheets Library

worksheets.clipart-library.com
Classifying Matter Worksheet With Answers – Classifying Matter

worksheets.clipart-library.com
PPT – Solution, Suspensions And Colloids PowerPoint Presentation, Free

www.slideserve.com
Solutions Colloids And Suspensions Worksheet – Artofit

www.artofit.org
Solutions Colloids And Suspensions Worksheet Ncert Exemplar Class 9

www.pinterest.com
Solutions Colloids And Suspensions Worksheet Classification Of Matters

www.pinterest.com
Solutions Colloids And Suspensions Worksheet – E-streetlight.com

www.e-streetlight.com
Solutions Colloids And Suspensions Worksheet – Printable PDF Template

martinlindelof.com
PPT – Suspensions And Colloids PowerPoint Presentation, Free Download
www.slideserve.com
Solutions Colloids And Suspensions Worksheet – Owhentheyanks.com
www.owhentheyanks.com
Solutions Colloids And Suspensions Worksheet – E-streetlight.com
www.e-streetlight.com
Solutions Colloids And Suspensions Worksheet – Ame.my.id

ame.my.id
Solutions Colloids And Suspensions Worksheet – Owhentheyanks.com
www.owhentheyanks.com
PPT – Suspensions And Colloids PowerPoint Presentation, Free Download
www.slideserve.com
solutions colloids and suspensions worksheet – e-streetlight.com. Solutions suspensions and colloids worksheet. solutions suspensions and colloids worksheet