Alright, science students! Let’s dive into the fascinating world of atoms! If you’ve been wrestling with your Atomic Theory Worksheet and are looking for a little guidance (or just a straight-up answer key, let’s be honest!), you’ve come to the right place. This isn’t just about giving you the answers, though. Understanding the *why* behind those answers is crucial for mastering the concepts. So, while we’ll provide the answers, we’ll also touch upon the key principles of atomic theory they relate to.
The atomic theory, which has been developed and refined over centuries by brilliant minds like Dalton, Thomson, Rutherford, and Bohr, is the foundation of all chemistry. It’s the framework that allows us to understand how matter is structured and how it behaves. From the simplest elements to the most complex molecules, everything boils down to the interactions of atoms. Getting a solid grasp of the basics now will pay dividends in your future chemistry studies.
Before we jump into the answers, remember that your worksheet might have variations depending on your teacher’s specific curriculum. Use these answers as a reference and always cross-reference them with your notes and textbook. It’s about learning, not just copying! And if you find any discrepancies or need further clarification, don’t hesitate to ask your teacher – they are there to help you!
Atomic Theory Worksheet Answers
Key Concepts Covered in the Worksheet:
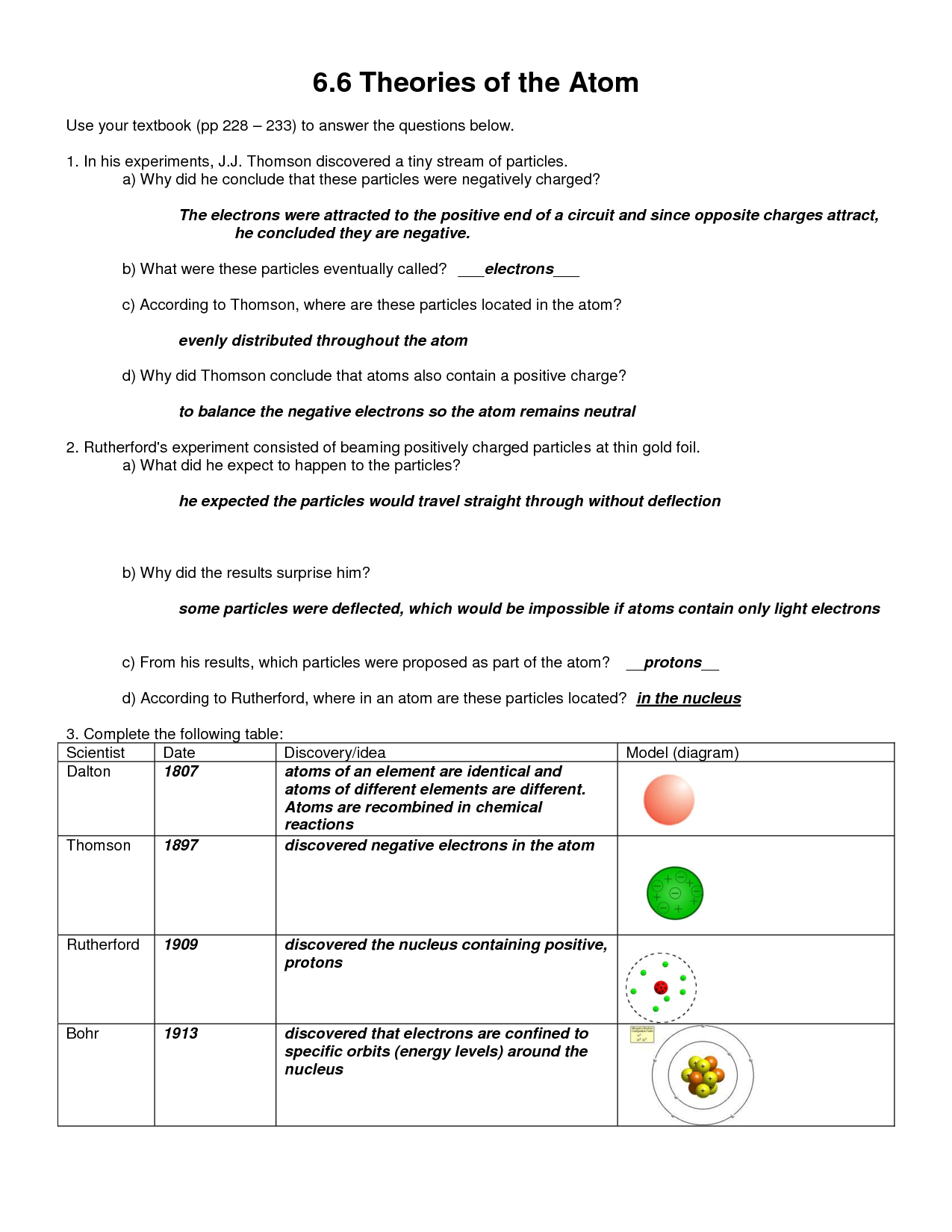
- Dalton’s Atomic Theory: The foundational principles about atoms being indivisible, elements having identical atoms, and compounds being formed by combinations of atoms in whole-number ratios.
- Thomson’s Plum Pudding Model: An early model of the atom suggesting a positively charged sphere with negatively charged electrons embedded within.
- Rutherford’s Gold Foil Experiment: Demonstrated the existence of a small, dense, positively charged nucleus.
- Bohr’s Atomic Model: Introduced the idea of electrons orbiting the nucleus in specific energy levels (shells).
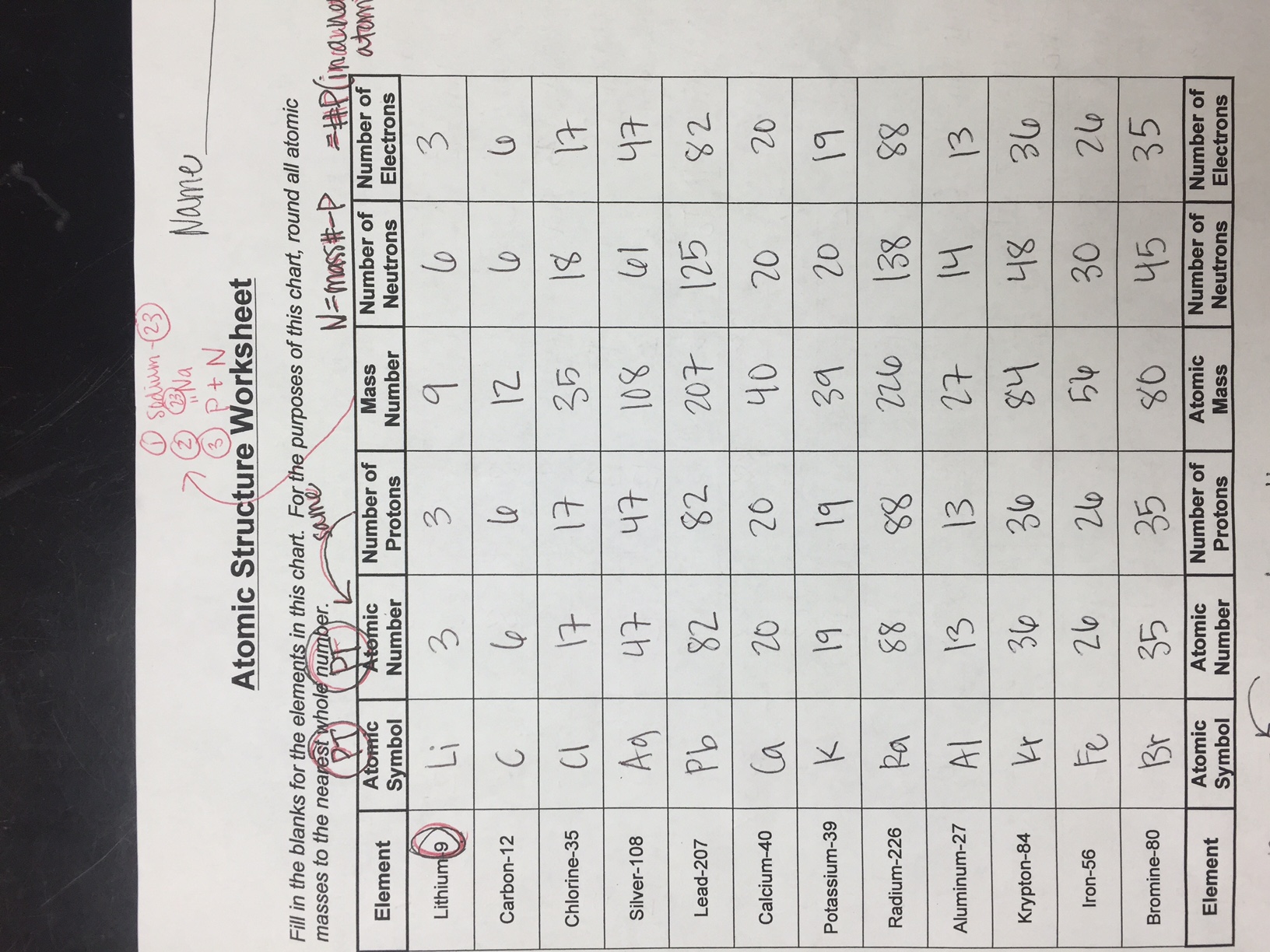
- Atomic Number and Mass Number: Understanding how these numbers relate to the number of protons, neutrons, and electrons in an atom.
- Isotopes and Ions: Differentiating between atoms of the same element with different numbers of neutrons (isotopes) and atoms that have gained or lost electrons (ions).
Now, let’s get to the answers! Below you’ll find a comprehensive list, formatted for clarity. Remember to use this as a tool to learn and understand the underlying concepts.
-
Question: What are the four main postulates of Dalton’s Atomic Theory?
- Answer:
- All matter is composed of extremely small particles called atoms.
- Atoms of a given element are identical in size, mass, and other properties; atoms of different elements differ in size, mass, and other properties.
- Atoms cannot be subdivided, created, or destroyed.
- Atoms of different elements combine in simple whole-number ratios to form chemical compounds.
- Answer:
-
Question: Describe Thomson’s “Plum Pudding” model of the atom.
- Answer: Thomson proposed that the atom was a positively charged sphere with negatively charged electrons scattered throughout, like plums in a pudding.
-
Question: Explain Rutherford’s gold foil experiment and what it revealed about the structure of the atom.
- Answer: Rutherford’s experiment involved firing alpha particles at a thin gold foil. Most particles passed straight through, but some were deflected at large angles. This showed that the atom is mostly empty space with a small, dense, positively charged nucleus.
-
Question: What did Bohr’s model of the atom contribute to our understanding of electron behavior?
- Answer: Bohr’s model introduced the idea that electrons orbit the nucleus in specific energy levels or shells. Electrons can only exist in these discrete energy levels, and they can jump between levels by absorbing or emitting energy.
-
Question: Define atomic number and mass number. How are they used to determine the number of protons, neutrons, and electrons in an atom?
- Answer:
- Atomic Number (Z): The number of protons in the nucleus of an atom. It uniquely identifies an element.
- Mass Number (A): The total number of protons and neutrons in the nucleus of an atom.
- Number of Protons = Atomic Number (Z)
- Number of Electrons = Atomic Number (Z) (in a neutral atom)
- Number of Neutrons = Mass Number (A) – Atomic Number (Z)
- Answer:
-
Question: What are isotopes? Give an example.
- Answer: Isotopes are atoms of the same element that have the same number of protons but different numbers of neutrons. For example, Carbon-12, Carbon-13, and Carbon-14 are all isotopes of carbon. They all have 6 protons, but they have 6, 7, and 8 neutrons respectively.
-
Question: What are ions? How do cations and anions differ?
- Answer: Ions are atoms or molecules that have gained or lost electrons, resulting in a net electrical charge.
- Cations: Positive ions formed when an atom loses electrons.
- Anions: Negative ions formed when an atom gains electrons.
-
Question: If an atom has 16 protons and 17 neutrons, what is its atomic number and mass number? What element is it?
- Answer:
- Atomic Number (Z) = 16
- Mass Number (A) = 16 + 17 = 33
- The element with atomic number 16 is Sulfur (S).
- Answer:
Remember, this is just a guide. Make sure to understand the reasoning behind each answer and relate it to the larger concepts of atomic theory. Good luck with your studies!
If you are searching about Worksheet Atomic Structure Answers – Owhentheyanks.com you’ve visit to the right web. We have 20 Pictures about Worksheet Atomic Structure Answers – Owhentheyanks.com like Atomic theory Worksheet Answers Fresh atomic theory Worksheet, Atomic Theory Worksheet Answers – E-streetlight.com and also WK Number 2 Atomic Structure Chemistry 1 Worksheet Assignment with. Read more:
Worksheet Atomic Structure Answers – Owhentheyanks.com

www.owhentheyanks.com
Atomic Structure Wkst – Science – Worksheet: Atomic Structure

worksheets.clipart-library.com
Atomic Structure Worksheet Pdf Best Of Atomic Structure Diagram

za.pinterest.com
Atomic Theory Worksheet – Alicia Barrett | Library | Formative

app.formative.com
Atomic Theory Worksheet Answers – E-streetlight.com

www.e-streetlight.com
Atomic Structure Worksheet Answers – E-streetlight.com

www.e-streetlight.com
Atoms And Atomic Structure – Worksheet | Printable And Distance

worksheets.clipart-library.com
Worksheet Atomic Theory – Atomic Theory Worksheet Name: Ashton Yates

www.studocu.com
Atomic Structure Worksheet 1 Answers
pcdhfvlessonmedia.z21.web.core.windows.net
Atomic Theory And Structure Of The Atom – Crossword Puzzle Worksheet

www.madebyteachers.com
SOLUTION: Chapter 02 Atomic Theory Element Properties Worksheet

worksheets.clipart-library.com
Worksheet Atomic Theory – Atomic Theory Worksheet Name Objective: You

www.studocu.com
Atomic Model Activity Worksheet

learningcampusornis.z13.web.core.windows.net
History Of Atomic Theory Worksheet – Printable Word Searches

davida.davivienda.com
Atomic Theory Worksheet Answers Pdf

classmagicpearce101.s3-website-us-east-1.amazonaws.com
History Of The Model Of The Atom Worksheet

learninglugnetlf.z21.web.core.windows.net
4.04 – Atomic Theory – Atomic Theory Worksheet Objective: You Will

worksheets.clipart-library.com
Atomic Theory Worksheet Answers – E-streetlight.com
www.e-streetlight.com
Atomic Theory Worksheet Answers Fresh Atomic Theory Worksheet

chessmuseum.org
WK Number 2 Atomic Structure Chemistry 1 Worksheet Assignment With

www.studocu.com
History of atomic theory worksheet. Solution: chapter 02 atomic theory element properties worksheet. Atoms and atomic structure