Struggling with double replacement reactions in chemistry? You’re not alone! These reactions, also known as metathesis reactions, can seem daunting at first. Predicting the products and determining whether a reaction will actually occur often requires careful consideration of solubility rules and understanding of driving forces. This post aims to help you master double replacement reactions with a helpful worksheet and a detailed answer key. We’ll break down the key concepts and give you the tools to confidently tackle these reactions. Think of this as your one-stop shop for conquering double replacement reactions!
Understanding Double Replacement Reactions
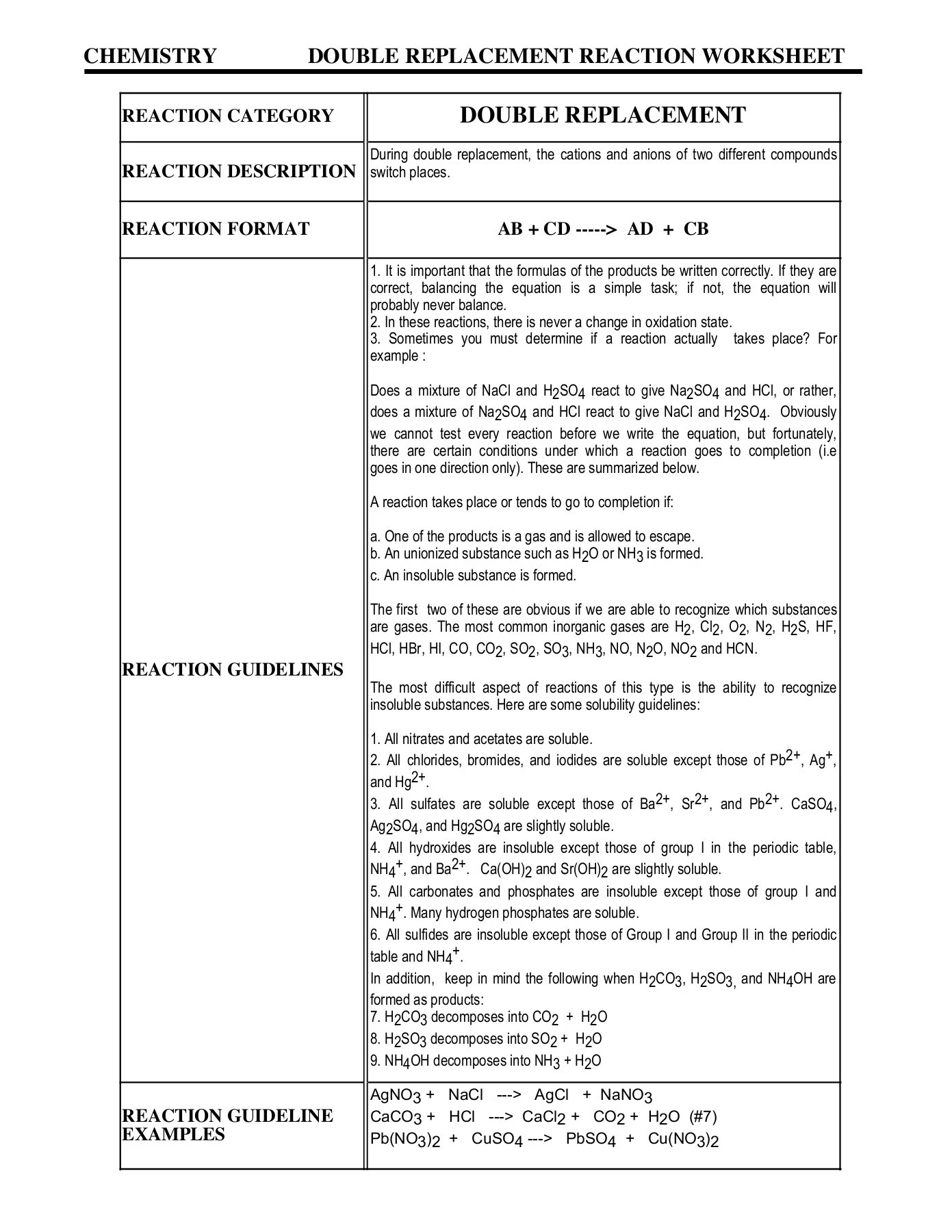
A double replacement reaction is a type of chemical reaction where two reactants exchange ions to form two new products. The general form of the reaction is:
AB + CD → AD + CB
Where A and C are cations (positively charged ions), and B and D are anions (negatively charged ions). The key to understanding these reactions is recognizing that they are driven by certain factors that favor the formation of stable products. These driving forces are:
- **Formation of a precipitate:** An insoluble solid (precipitate) forms from the reaction of two aqueous solutions.
- **Formation of a gas:** A gas is produced as one of the products.
- **Formation of a molecular compound, such as water:** The reaction results in the formation of a stable, non-ionized molecule, typically water.
If none of these driving forces are present, the reaction will likely not occur to a significant extent. Therefore, predicting whether a double replacement reaction will occur involves knowing your solubility rules and being able to identify common gases and molecular compounds formed in these types of reactions.
Tips for Balancing Double Replacement Reactions
Balancing chemical equations is crucial for accurately representing the stoichiometry of a reaction. Here are a few tips to keep in mind when balancing double replacement reactions:
- **Write the correct formulas:** Ensure you correctly identify the ions and use the proper charges to create neutral compounds. Double-check your work; incorrect formulas will lead to incorrect balancing.
- **Start with complex compounds:** Begin balancing with the compounds that have the most atoms. This can often simplify the process.
- **Balance polyatomic ions as a unit:** If a polyatomic ion appears on both sides of the equation unchanged, treat it as a single unit during balancing. This significantly reduces the steps required.
- **Check your work:** After balancing, carefully count the number of atoms of each element on both sides of the equation to ensure they are equal.
Double Replacement Reaction Worksheet Answers
Here are the answers to a typical double replacement reaction worksheet. Remember, the key is to correctly identify the products and then balance the equation. Also, don’t forget to indicate the state of matter (aqueous, solid, gas, liquid) for each product!
-
Question: Silver nitrate (AgNO3) reacts with sodium chloride (NaCl).
Answer:- Balanced Equation: AgNO3(aq) + NaCl(aq) → AgCl(s) + NaNO3(aq)
-
Question: Barium chloride (BaCl2) reacts with sodium sulfate (Na2SO4).
Answer:- Balanced Equation: BaCl2(aq) + Na2SO4(aq) → BaSO4(s) + 2NaCl(aq)
-
Question: Lead(II) nitrate (Pb(NO3)2) reacts with potassium iodide (KI).
Answer:- Balanced Equation: Pb(NO3)2(aq) + 2KI(aq) → PbI2(s) + 2KNO3(aq)
-
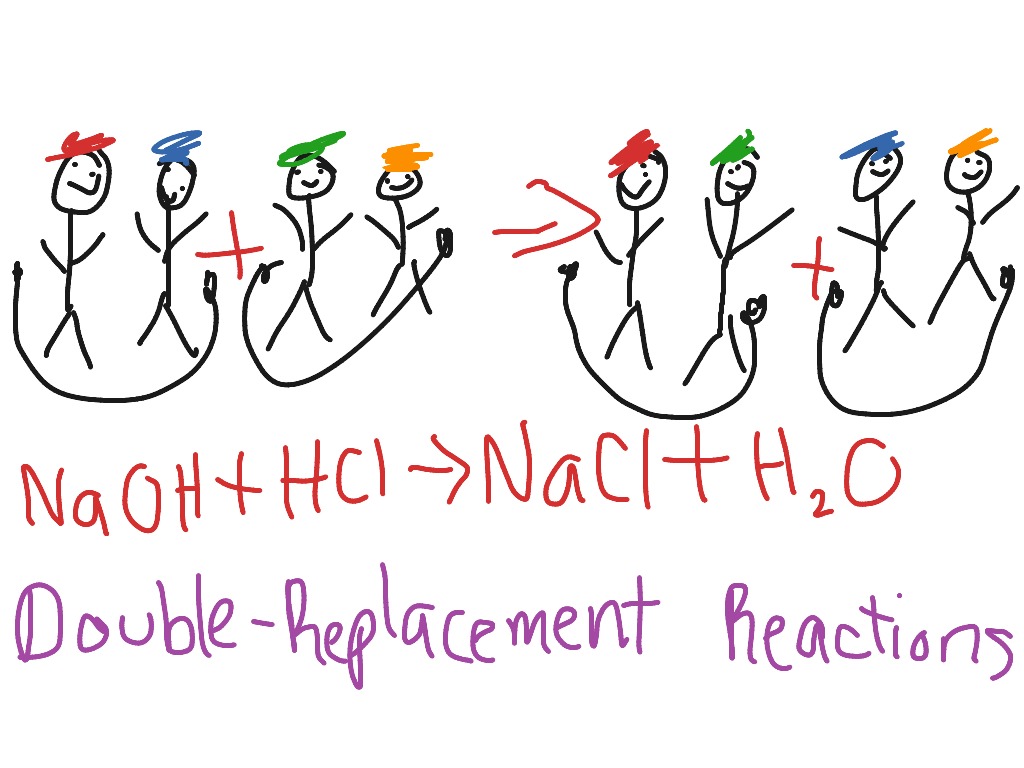
Question: Sodium hydroxide (NaOH) reacts with hydrochloric acid (HCl).
Answer:- Balanced Equation: NaOH(aq) + HCl(aq) → H2O(l) + NaCl(aq)
-
Question: Potassium carbonate (K2CO3) reacts with nitric acid (HNO3).
Answer:- Balanced Equation: K2CO3(aq) + 2HNO3(aq) → 2KNO3(aq) + H2O(l) + CO2(g)
-
Question: Ammonium sulfide ((NH4)2S) reacts with iron(III) nitrate (Fe(NO3)3).
Answer:- Balanced Equation: 3(NH4)2S(aq) + 2Fe(NO3)3(aq) → Fe2S3(s) + 6NH4NO3(aq)
-
Question: Magnesium chloride (MgCl2) reacts with sodium phosphate (Na3PO4).
Answer:- Balanced Equation: 3MgCl2(aq) + 2Na3PO4(aq) → Mg3(PO4)2(s) + 6NaCl(aq)
-
Question: Copper(II) sulfate (CuSO4) reacts with sodium hydroxide (NaOH).
Answer:- Balanced Equation: CuSO4(aq) + 2NaOH(aq) → Cu(OH)2(s) + Na2SO4(aq)
-
Question: Strontium chloride (SrCl2) reacts with potassium chromate (K2CrO4).
Answer:- Balanced Equation: SrCl2(aq) + K2CrO4(aq) → SrCrO4(s) + 2KCl(aq)
-
Question: Zinc bromide (ZnBr2) reacts with ammonium carbonate ((NH4)2CO3).
Answer:- Balanced Equation: ZnBr2(aq) + (NH4)2CO3(aq) → ZnCO3(s) + 2NH4Br(aq)
By understanding the principles behind double replacement reactions, practicing with worksheets, and reviewing the answers, you’ll be well on your way to mastering this important concept in chemistry. Good luck!
If you are looking for SOLUTION: Single Replacement Reaction Worksheet answer key – Studypool you’ve visit to the right page. We have 20 Pics about SOLUTION: Single Replacement Reaction Worksheet answer key – Studypool like Chemistry Single Replacement Reaction Worksheet: L.Ag + Kno | PDF, Double Replacement Reaction Worksheet – Pro Worksheet and also Double Replacement Reaction Worksheet – E-streetlight.com. Here you go:
SOLUTION: Single Replacement Reaction Worksheet Answer Key – Studypool

worksheets.clipart-library.com
Topic-6-Predicting Single Replacement Reactions-cglass | St

worksheets.clipart-library.com
Double Replacement Reaction Worksheet – E-streetlight.com

www.e-streetlight.com
Chemistry Single Replacement Reaction Worksheet: L.Ag + Kno | PDF
worksheets.clipart-library.com
Double Replacement Reaction Worksheet – Pro Worksheet

www.proworksheet.my.id
Double Replacement Reaction Worksheet – Pro Worksheet

www.proworksheet.my.id
Double Displacement Reactions Worksheet – Worksheets For Kindergarten

worksheets.ekocraft-appleleaf.com
SOLUTION: Single Replacement Reaction Worksheet Answer Key – Studypool

worksheets.clipart-library.com
Single Replacement Reaction Worksheets

quizzschooldawkins.z21.web.core.windows.net
Free Single Replacement Reaction Worksheets For Students – Worksheets

worksheets.clipart-library.com
Worksheet #4: Single-Replacement Reactions – ScienceGeek.net

worksheets.clipart-library.com
Double Replacement Reaction Worksheet – E-streetlight.com

www.e-streetlight.com
Double Displacement Reactions Worksheet

lessoncampusford.z13.web.core.windows.net
Synthesis Reactions And Decomposition Reactions Worksheet Key

worksheets.clipart-library.com
Worksheet On Single Double Replacement Reactions – Worksheets For
worksheets.ekocraft-appleleaf.com
Double Replacement Reaction Worksheet | Reaction Types, Chemistry

www.pinterest.com
Double Replacement Example
.jpg)
ar.inspiredpencil.com
Double Replacement Reaction Worksheet – Pro Worksheet

www.proworksheet.my.id
A Double-displacement Reaction(also Called A | Chegg.com
www.chegg.com
Double Replacement Reaction Worksheet – E-streetlight.com

www.e-streetlight.com
Single replacement reaction worksheets. Synthesis reactions and decomposition reactions worksheet key. double replacement reaction worksheet – pro worksheet
