Ah, the dreaded Acid and Base Worksheet! Whether you’re a high school chemistry student just diving into the world of pH scales and titrations, or a college student refreshing your knowledge for a more advanced course, understanding acids and bases is absolutely fundamental to grasping countless chemical principles. But let’s face it: working through those worksheets can be a real challenge. Understanding the concepts is one thing, but applying them to solve problems and correctly identify acids, bases, conjugates, and all those other intricate details requires practice and, sometimes, a little bit of help. This post aims to provide just that – a comprehensive guide to help you understand your acid-base worksheet answers and, more importantly, understand *why* those answers are correct. Remember, it’s not just about memorizing; it’s about understanding the underlying chemistry. Let’s dive in!
Understanding the Core Concepts
Before we even touch the answers, let’s quickly recap the key concepts. Understanding these will make deciphering the solutions much easier and prevent you from just blindly copying down answers. We need to talk about:
- **Acids:** Substances that donate protons (H+) or accept electrons. They typically have a sour taste (though you shouldn’t be tasting them in the lab!) and can react with metals.
- **Bases:** Substances that accept protons (H+) or donate electrons. They typically have a bitter taste and feel slippery.
- **pH Scale:** A logarithmic scale that measures the acidity or basicity of a solution. Values range from 0 to 14, with 7 being neutral (like pure water). pH values below 7 indicate acidity, while values above 7 indicate basicity.
- **Strong Acids and Bases:** These completely dissociate in water, meaning they break apart into their ions. Examples of strong acids include hydrochloric acid (HCl), sulfuric acid (H2SO4), and nitric acid (HNO3). Examples of strong bases include sodium hydroxide (NaOH) and potassium hydroxide (KOH).
- **Weak Acids and Bases:** These only partially dissociate in water. This means that not all of the acid or base molecules break apart into ions. They reach an equilibrium between the undissociated form and the ions. Examples of weak acids include acetic acid (CH3COOH) and hydrofluoric acid (HF). Examples of weak bases include ammonia (NH3) and pyridine (C5H5N).
- **Conjugate Acids and Bases:** When an acid donates a proton, the remaining species is its conjugate base. When a base accepts a proton, the resulting species is its conjugate acid. For example, in the reaction HCl + H2O → H3O+ + Cl–, Cl– is the conjugate base of HCl, and H3O+ is the conjugate acid of H2O.
- **Neutralization Reactions:** The reaction between an acid and a base, typically forming a salt and water.
Sample Acid And Base Worksheet Answers
Here are some example questions you might find on a typical acid-base worksheet, along with their corresponding answers and explanations. Remember, your worksheet questions might be different, but understanding these examples will give you a solid foundation.
Example Questions and Answers
-
Question: Identify the acid, base, conjugate acid, and conjugate base in the following reaction: NH3(aq) + H2O(l) ⇌ NH4+(aq) + OH–(aq)
Answer:
- Acid: H2O
- Base: NH3
- Conjugate Acid: NH4+
- Conjugate Base: OH–
Explanation: NH3 accepts a proton (H+) from H2O, making it the base. H2O donates a proton, making it the acid. NH4+ is formed when NH3 accepts the proton, so it’s the conjugate acid. OH– is formed when H2O donates the proton, so it’s the conjugate base.
-
Question: Calculate the pH of a 0.01 M solution of HCl.
Answer: pH = 2
Explanation: HCl is a strong acid, so it completely dissociates in water. This means that the concentration of H+ ions is equal to the concentration of HCl, which is 0.01 M. The pH is calculated using the formula: pH = -log[H+]. Therefore, pH = -log(0.01) = 2. -
Question: Identify the following compounds as acids, bases, or neither: NaOH, HBr, CH3COOH, NaCl.
Answer:
- NaOH: Base
- HBr: Acid
- CH3COOH: Acid
- NaCl: Neither
Explanation: NaOH is a strong base (sodium hydroxide). HBr is a strong acid (hydrobromic acid). CH3COOH is a weak acid (acetic acid). NaCl is a salt formed from the reaction of a strong acid (HCl) and a strong base (NaOH), so it is neither acidic nor basic in a simple aqueous solution (it’s pH neutral, assuming perfect neutrality of the water used).
-
Question: What is the conjugate base of H2SO4?
Answer: HSO4–
Explanation: The conjugate base is formed by removing a proton (H+) from the acid. Removing one H+ from H2SO4 leaves HSO4–.
Importance of Practice
The key to mastering acid-base chemistry, and understanding your acid and base worksheet answers, is consistent practice. Work through multiple problems, focusing on the underlying concepts rather than just memorizing formulas. Pay attention to the stoichiometry of the reactions, the strengths of the acids and bases, and the relationships between conjugate acid-base pairs. If you’re struggling with a particular type of problem, seek out additional resources, such as online tutorials, textbook examples, or help from your teacher or professor. Remember, understanding acids and bases is crucial not only for chemistry but also for many other scientific fields, including biology, medicine, and environmental science.
If you are searching about Acid, Bases, Salts – IGCSE Chemistry (solutions, examples you’ve came to the right place. We have 20 Pictures about Acid, Bases, Salts – IGCSE Chemistry (solutions, examples like Acid And Base Worksheet Answers – Pro Worksheet, Acid / Base Balance Worksheet – Worksheets Library and also Acid base worksheet | PDF. Here you go:
Acid, Bases, Salts – IGCSE Chemistry (solutions, Examples

worksheets.clipart-library.com
Acid / Base Balance Worksheet – Worksheets Library

worksheets.clipart-library.com
Acid Base Calculations Worksheet

lessonschoolcheckmates.z14.web.core.windows.net
Copy Of Acid Base Worksheet.docx – SCH4U ACID-BASE WORKSHEET Complete

www.studocu.com
Acid Base Worksheet Answers – CHEM 101 – Studocu – Worksheets Library

worksheets.clipart-library.com
SOLUTION: Acids And Bases Worksheet – Studypool – Worksheets Library

worksheets.clipart-library.com
Acid And Base Theories Worksheet

learningzoneplanmatigic.z21.web.core.windows.net
Acid And Base Worksheet Answers – Pro Worksheet

www.proworksheet.my.id
Acid Base Worksheet – Homework – Acid Base Worksheet Name

www.studocu.com
Acid Base Practice Questions Nursing

worksheetukletognk.z21.web.core.windows.net
Titration Practice Acid Base Reaction Worksheet With Answer Key

worksheets.clipart-library.com
Acid And Base Worksheet – E-streetlight.com
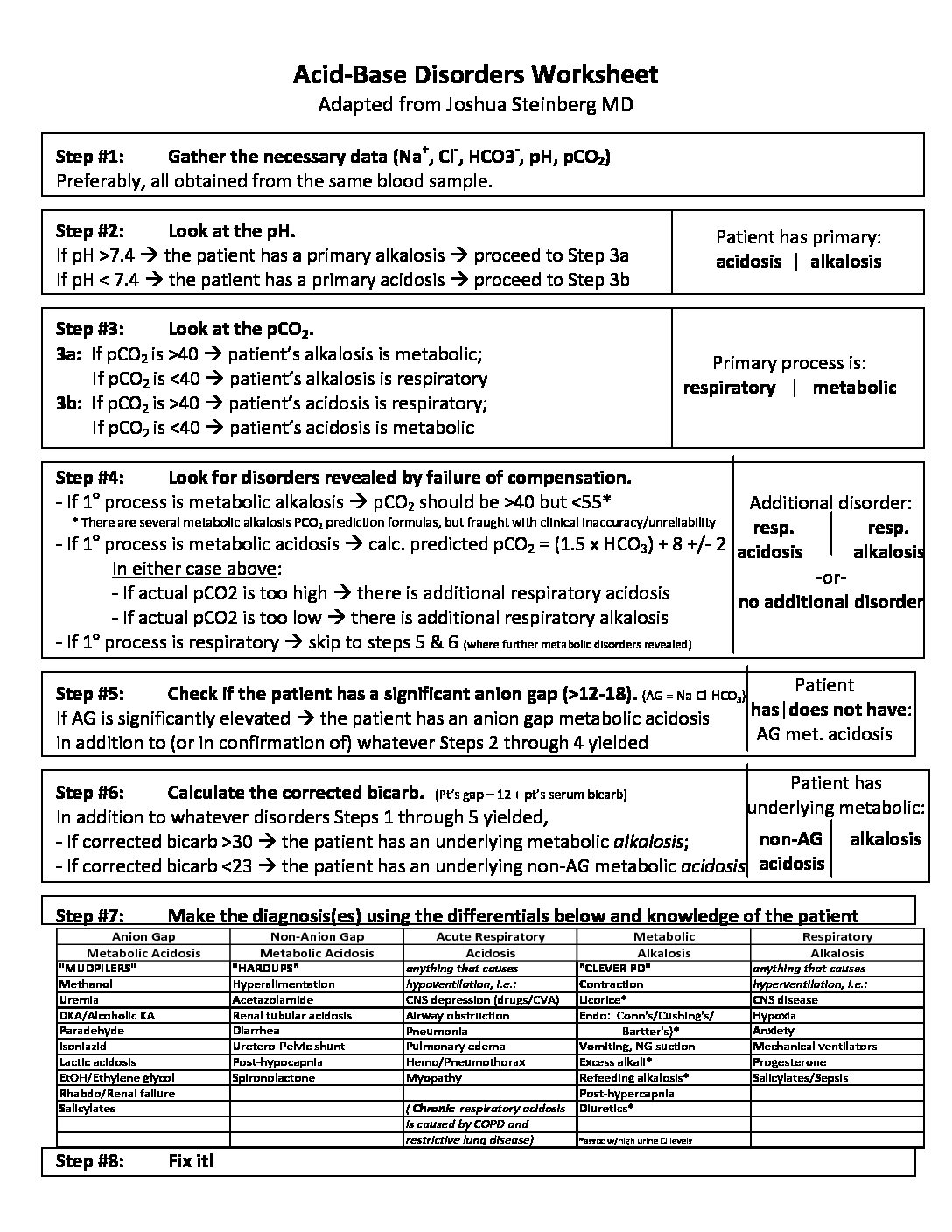
www.e-streetlight.com
Acid Base Worksheet | PDF

www.slideshare.net
Acid Base Titration Worksheet Answers Acid Base Titration Worksheet

www.aiophotoz.com
Acid And Base Worksheet – E-streetlight.com

www.e-streetlight.com
Titration Practice Worksheet Answers – Printable PDF Template

martinlindelof.com
Acid-Base Equilibria – Worksheet 16 With Answers | CHEM 116

worksheets.clipart-library.com
Acid Base Worksheet Student COPY (2015 ) – Acids & Bases Fill In The
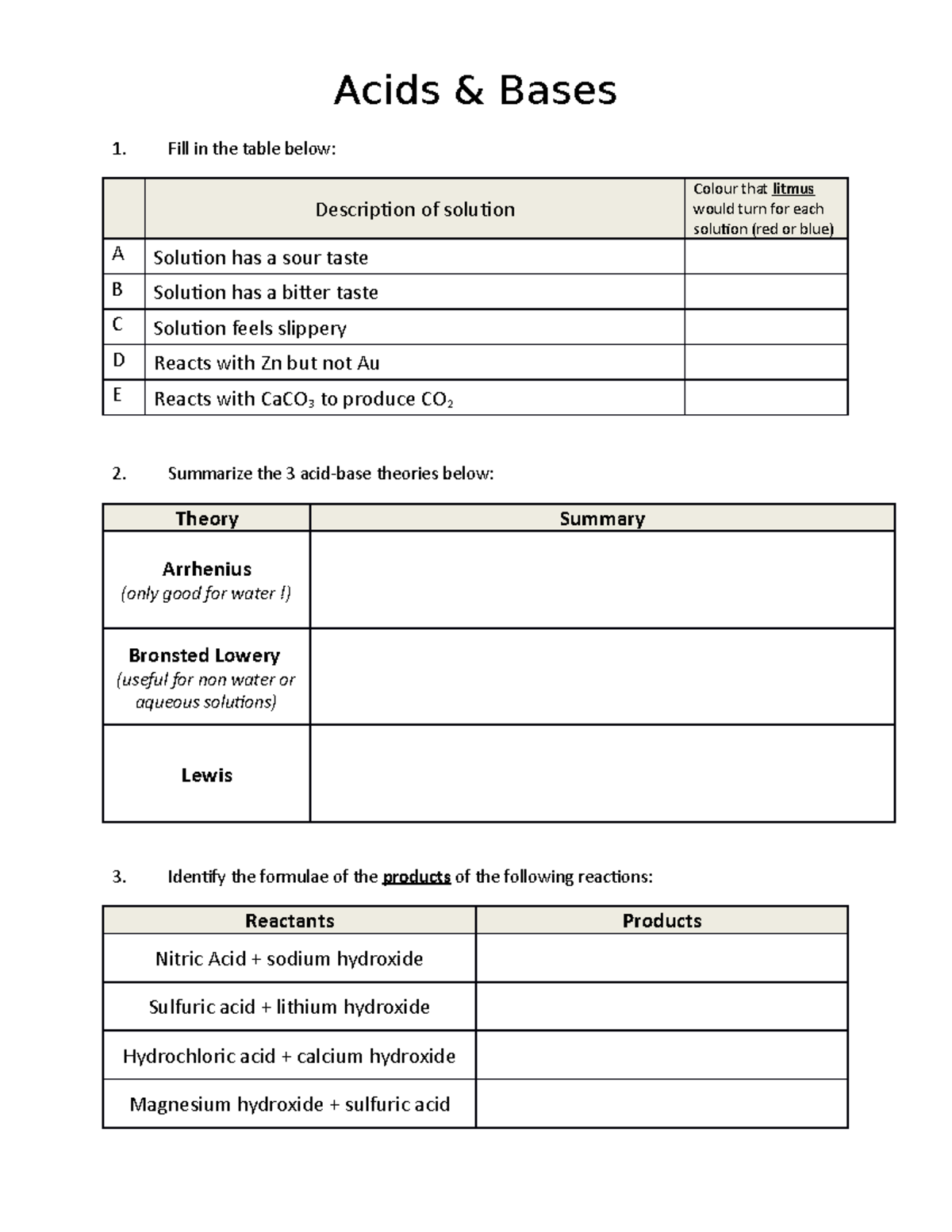
www.studocu.com
Phet Acid Base Solutions Worksheet Answers

ideilorjljlessonlearning.z14.web.core.windows.net
Acid Base Worksheet – Acid Base – Critical Thinking Worksheet

worksheets.clipart-library.com
Titration practice acid base reaction worksheet with answer key …. acid base worksheet. Acid base worksheet