Understanding the behavior of gases is fundamental to comprehending many scientific principles, from weather patterns to the inner workings of engines. These principles are governed by several laws that describe the relationships between pressure, volume, temperature, and the number of moles of gas. Worksheets focusing on the behavior of gases offer a fantastic way to practice applying these laws and solidifying your understanding. This post will delve into the key concepts covered in a typical “Behavior of Gases” worksheet and then provide example answers to common types of questions. Get ready to sharpen your skills and boost your confidence in gas laws!
Key Concepts Covered in a Behavior of Gases Worksheet
A comprehensive “Behavior of Gases” worksheet typically covers the following core concepts and gas laws:
* **Boyle’s Law:** This law states that for a fixed amount of gas at a constant temperature, the pressure and volume are inversely proportional. Mathematically, this is expressed as P₁V₁ = P₂V₂. Essentially, if you compress a gas (decrease its volume), its pressure increases, and vice versa.
* **Charles’s Law:** This law states that for a fixed amount of gas at constant pressure, the volume is directly proportional to the absolute temperature (in Kelvin). The formula is V₁/T₁ = V₂/T₂. Heating a gas will cause it to expand. It’s crucial to remember to always use Kelvin for temperature calculations.
* **Gay-Lussac’s Law:** This law states that for a fixed amount of gas at constant volume, the pressure is directly proportional to the absolute temperature. The formula is P₁/T₁ = P₂/T₂. Increase the temperature of a gas in a closed container, and the pressure will increase.
* **Avogadro’s Law:** This law states that equal volumes of all gases, at the same temperature and pressure, contain the same number of molecules. This leads to the concept of molar volume (22.4 L/mol at standard temperature and pressure, or STP).
* **Ideal Gas Law:** This law combines Boyle’s, Charles’s, Gay-Lussac’s, and Avogadro’s laws into a single equation: PV = nRT, where P is pressure, V is volume, n is the number of moles, R is the ideal gas constant, and T is temperature in Kelvin.
* **Combined Gas Law:** This law combines Boyle’s, Charles’s, and Gay-Lussac’s laws into a single equation: (P₁V₁)/T₁ = (P₂V₂)/T₂. It is useful when all three variables (pressure, volume, and temperature) are changing.
* **Dalton’s Law of Partial Pressures:** This law states that the total pressure exerted by a mixture of gases is equal to the sum of the partial pressures of each individual gas. Ptotal = P₁ + P₂ + P₃ + …
* **Gas Stoichiometry:** Using the ideal gas law to relate the volume of a gas to the number of moles involved in a chemical reaction.
Worksheets will often present problems that require you to identify which law applies, rearrange the formula, and solve for the unknown variable. They may also involve unit conversions (e.g., Celsius to Kelvin, mmHg to atm) and stoichiometric calculations.
Example Problems and Solutions
Below are example questions you might find on a “Behavior of Gases” worksheet, along with their solutions. Remember to always show your work and include the correct units!
- Question 1: A gas occupies a volume of 10.0 L at standard temperature and pressure (STP). What volume will it occupy at 2.0 atm and 273°C?
- Question 2: A balloon contains 5.0 L of gas at 25°C and 1 atm. If the temperature is increased to 50°C and the pressure remains constant, what is the new volume of the balloon?
- Question 3: A container holds 4.0 g of helium gas at 27°C and a pressure of 600 torr. What is the volume of the container?
- Question 4: A mixture of gases contains 2.0 moles of nitrogen gas and 3.0 moles of oxygen gas. If the total pressure of the mixture is 3.0 atm, what is the partial pressure of the nitrogen gas?
Answers to Example Problems
Here are the solutions to the example problems. Remember to pay close attention to units and significant figures!
- Answer 1:
We can use the combined gas law: (P₁V₁)/T₁ = (P₂V₂)/T₂
- P₁ = 1 atm
- V₁ = 10.0 L
- T₁ = 273 K (STP)
- P₂ = 2.0 atm
- T₂ = 273 + 273 = 546 K
- V₂ = ?
(1 atm * 10.0 L) / 273 K = (2.0 atm * V₂) / 546 K
V₂ = (1 atm * 10.0 L * 546 K) / (273 K * 2.0 atm) = 10.0 L
Therefore, the volume at 2.0 atm and 273°C is 10.0 L.
- Answer 2:
We can use Charles’s Law: V₁/T₁ = V₂/T₂
- V₁ = 5.0 L
- T₁ = 25 + 273 = 298 K
- T₂ = 50 + 273 = 323 K
- V₂ = ?
5.0 L / 298 K = V₂ / 323 K
V₂ = (5.0 L * 323 K) / 298 K = 5.4 L
Therefore, the new volume of the balloon is approximately 5.4 L.
- Answer 3:
We can use the Ideal Gas Law: PV = nRT
- P = 600 torr = 600/760 atm ≈ 0.789 atm
- V = ?
- n = 4.0 g He / 4.0 g/mol ≈ 1.0 mol
- R = 0.0821 L atm / (mol K)
- T = 27 + 273 = 300 K
0. 789 atm * V = 1.0 mol * 0.0821 L atm / (mol K) * 300 K
V = (1.0 mol * 0.0821 L atm / (mol K) * 300 K) / 0.789 atm ≈ 31.2 L
Therefore, the volume of the container is approximately 31.2 L.
- Answer 4:
We can use Dalton’s Law of Partial Pressures and the concept of mole fraction.
Mole fraction of N₂ = 2.0 mol / (2.0 mol + 3.0 mol) = 0.4
P(N₂) = Mole fraction of N₂ * Ptotal
P(N₂) = 0.4 * 3.0 atm = 1.2 atm
Therefore, the partial pressure of the nitrogen gas is 1.2 atm.
If you are searching about Printable Home School Science Worksheet Graphic by Island Tulip you’ve came to the right web. We have 20 Images about Printable Home School Science Worksheet Graphic by Island Tulip like Behavior Of Gases Worksheet Beautiful Quiz & Worksheet the Outsiders, Agenda Behavior of Gases Gas Laws Gas Laws Worksheet. – ppt download and also Kinetic Molecular Theory of Matter Chemistry Activities. Here you go:
Printable Home School Science Worksheet Graphic By Island Tulip

worksheets.clipart-library.com
Behavior Of Gases Worksheet – E-streetlight.com

www.e-streetlight.com
Solids, Liquids, Gases Lesson Plan Grade 2 – Worksheets Library

worksheets.clipart-library.com
Behavior Of Gases Worksheet Beautiful Quiz & Worksheet The Outsiders
chessmuseum.org
Behavior Think Sheet Worksheets – Worksheets Library

worksheets.clipart-library.com
Behavior Of Gases Worksheet – E-streetlight.com

www.e-streetlight.com
Lab 3a Pdf Exploring The Behaviour Of Gases – Exploring The

worksheets.clipart-library.com
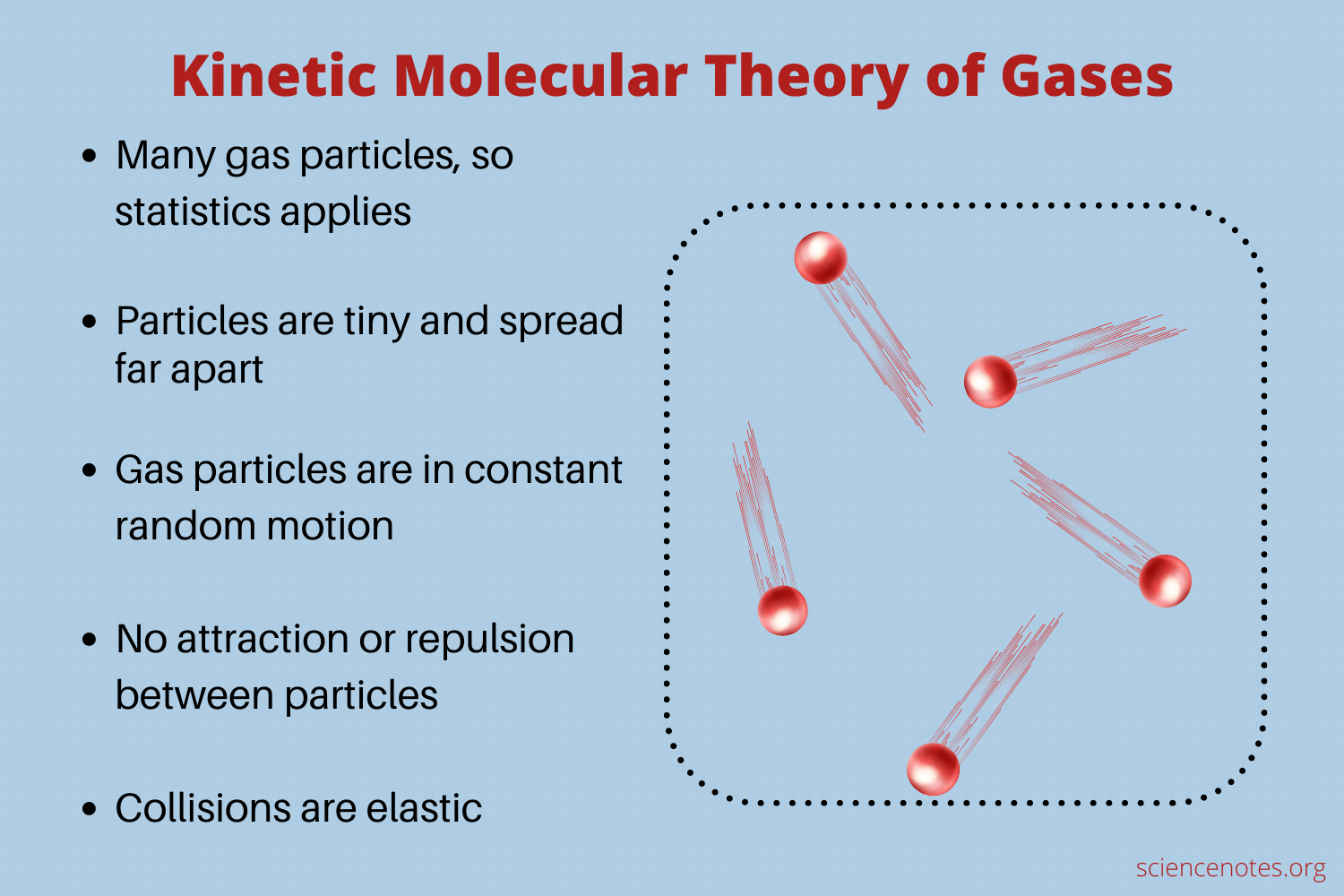
Kinetic Molecular Theory Of Gases – Worksheets Library
worksheets.clipart-library.com
Free Homophones Worksheets – For Kids And Classroom – Worksheets Library

worksheets.clipart-library.com
Cup Of Feelings Worksheet – Etsy – Worksheets Library

worksheets.clipart-library.com
Behavior Of Gases Worksheet – E-streetlight.com

www.e-streetlight.com
Free, Custom Printable Worksheet Templates For Teachers | Canva

worksheets.clipart-library.com
SOLUTION: General Chemistry General And Measurable Properties Of

worksheets.clipart-library.com
Electron Configuration Worksheet Answers Worksheet Ii Electron

worksheets.clipart-library.com
Weathering – Physical, Chemical, Biological – Worksheets Library

worksheets.clipart-library.com
Kinetic Molecular Theory Of Matter Chemistry Activities

worksheets.clipart-library.com
Agenda Behavior Of Gases Gas Laws Gas Laws Worksheet. – Ppt Download

worksheets.clipart-library.com
Behavior Of Gases Worksheet – Pro Worksheet

www.proworksheet.my.id
SOLUTION: Ideal Gass Law Worksheet – Studypool – Worksheets Library

worksheets.clipart-library.com
Physical And Chemical Changes – Worksheets Library

worksheets.clipart-library.com
behavior think sheet worksheets. Cup of feelings worksheet. Behavior of gases worksheet – e-streetlight.com